
Page 31
http://threesology.org
Note: the contents of this page as well as those which precede and follow, must be read as a continuation and/or overlap in order that the continuity about a relationship to/with the typical dichotomous assignment of Artificial Intelligence (such as the usage of zeros and ones used in computer programming) as well as the dichotomous arrangement of the idea that one could possibly talk seriously about peace from a different perspective... will not be lost (such as war being frequently used to describe an absence of peace and vice-versa). However, if your mind is prone to being distracted by timed or untimed commercialization (such as that seen in various types of American-based television, radio, news media and magazine publishing... not to mention the average classroom which carries over into the everyday workplace), you may be unable to sustain prolonged exposures to divergent ideas about a singular topic without becoming confused, unless the information is provided in a very simplistic manner.
Note: when we think of computer speed, we are thinking in terms of ability to process, or move quickly. To be able to sustain such activity over long periods without fatiguing components is of crucial importance in many applications. How a computer heats up, breaths and can refresh itself is comparable to these same features in animate systems. Do we need to alter the type of blood, food, "heart rate" and "lung capacity" to achieve our goals at creating an advanced AI system? I am not speaking in a literal sense, but a metaphorical one... (said for those readers who do not have the capacity to change intellectual gears very quickly due to being presented with different terrains of subject matter).
Modern reptiles do not have the capacity for the rapid sustained activity found in birds and mammals. It is generally accepted that this lower capacity is related to differences in the circulatory and respiratory systems. Before the origin of lungs, the vertebrate circulatory system had a single circuit: in the fishes, blood flows from heart to gills to body and back to the heart. The heart consists of four chambers arranged in a linear sequence. With the evolution of lungs in early tetrapods, a new and apparently more efficient circulatory system evolved. Two chambers of the heart, the atrium (or auricle) and ventricle, became increasingly important, and the beginnings of double circulation appeared. An early stage in this evolution can be seen in amphibians today, where one of the main arteries from the heart (the pulmonary artery) goes directly to the lungs, whereas the others (the systemic arteries) carry blood to the general body. In amphibians the blood is aerated in the lungs and carried back to the atrium of the heart. From the left side of the atrium, which is at least partially divided, the aerated blood is pumped into the ventricle to mix with nonaerated blood; nonaerated blood from the body is returned to the heart via the right half of the atrium. Then the cycle begins again. One aspect of the amphibian system is that the blood leaving the heart for the body is only partially aerated; part of it is made up of deoxygenated blood returned from the body. All groups of modern reptiles have a completely divided atrium; it is safe to assume, therefore, that this was true of most, if not all, extinct reptiles. In the four major living groups of reptiles, the ventricle is at least partially divided. When the two atria of a lizard's heart contract, the two streams of blood (aerated blood from the lungs in the left atrium and nonaerated blood from the body in the right atrium) flow into the left chamber of the ventricle. As pressure builds up in that chamber, the nonaerated blood is forced through the gap in the partition into the right chamber of the ventricle. Then, when the ventricle contracts, nonaerated blood is pumped into the pulmonary artery and thence to the lungs, whereas aerated blood is pumped into the systemic arteries (the aortas) and so to the body. In snakes all three arterial trunks come out of the chamber of the ventricle that receives the nonaerated blood of the right atrium. During ventricular contraction a muscular ridge forms a partition that guides the nonaerated blood into the pulmonary artery, while the aerated blood received by the other chamber of the ventricle is forced through the opening in the ventricular septum and out through the aortas. In crocodiles the ventricular septum is complete, but the two aortas come out of different ventricular chambers. A semilunar valve at the entrance to the left aorta prevents nonaerated blood in the right ventricle from flowing into the aorta. Instead, part of the aerated blood from the left ventricular chamber pumped into the right aorta flows into the left by way of an opening. The ventricle of the turtle is not perfectly divided, and some slight mixing of aerated and nonaerated blood can occur. Despite the peculiar and complex circulation, lizards, snakes, and crocodilians have achieved a double system. Tests of the blood in the various chambers and arteries have shown that the oxygen content in both systemic aortas is as high as that of the blood just received by the left atrium from the lungs and is much higher than that of the blood in the pulmonary artery. Except for the turtles, limitation of activity in reptiles cannot be explained on the basis of heart circulation. An explanation may lie in the chemistry of the blood. The blood of reptiles has less hemoglobin and thus carries less oxygen than that of mammals. Source: "Reptile." Encyclopædia Britannica Ultimate Reference Suite, 2013. |
With respect to the movement of fluid, though swimming techniques are well recognized, they are not typically referred to as valve systems... even though they are concerned with the displacement of water from front to rear of a swimmer. Nonetheless, despite the objections some readers may want to invoke, let me provide a few illustrated examples of swimming containing names such as side stroke, back stroke, front (freestyle) stroke, butterfly stroke, breast stroke, "flipper stroke" (use of flippers only), dog paddle, etc... (As it had been said: Different strokes for different folks.)
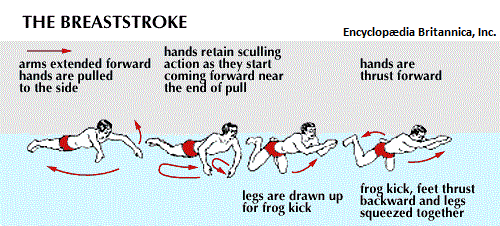 |
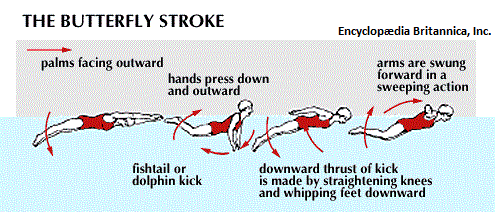 |
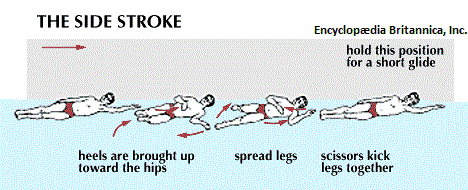
| |
In an application of thinking in terms of electronic circuitry, the swimmer can be viewed as a single electron particle with different movements due to variations of environmental exposure to a given "medium" (such as element used in the construction of an electrical component). Each style of swimming is a binary rhythm that may or may not synchronize the use of the legs and breathing/breath holding movements. While most athletes train vigorously while consuming a prescribed diet of food, beverage, sleep, relaxation as well as vitamins and minerals, some engage in what is described as "doping", which is the usage of steriods to enhance performance. While it is frowned on and even outlawed in many sports venues, because it is viewed as both an artificiality of ability and medically unsafe in many circumstances; in the world of electronics, the use of doping materials has been found to be advantageous... in an effort to seek out the development of superconductors that at present require extreme environments such as a greatly lowered temperature setting, sometimes achieved by the usage of liquid nitrogen. (Recall that Nitrogen is a constituent of all cells, and the usage of nitrogen in a liquid form nonetheless intimates a biological direction involving computer development.)
Doping silicon Most ICs (integrated circuits) are made of silicon, which is abundant in ordinary beach sand. Pure crystalline silicon, as with other semiconducting materials, has a very high resistance to electrical current at normal room temperature. However, with the addition of certain impurities, known as dopants, the silicon can be made to conduct usable currents. In particular, the doped silicon can be used as a switch, turning current off and on as desired. The process of introducing impurities is known as doping or implantation. Depending on a dopant's atomic structure, the result of implantation will be either an n-type (negative) or a p-type (positive) semiconductor. An n-type semiconductor results from implanting dopant atoms that have more electrons in their outer (bonding) shell than silicon, as shown in the figure. The resulting semiconductor crystal contains excess, or free, electrons that are available for conducting current. A p-type semiconductor results from implanting dopant atoms that have fewer electrons in their outer shell than silicon. The resulting crystal contains “holes” in its bonding structure where electrons would normally be located. In essence, such holes can move through the crystal conducting positive charges. A p-type or an n-type semiconductor is not very useful on its own. However, joining these opposite materials creates what is called a p-n junction (see figure). A p-n junction forms a barrier to conduction between the materials. Although the electrons in the n-type material are attracted to the holes in the p-type material, the electrons are not normally energetic enough to overcome the intervening barrier. However, if additional energy is provided to the electrons in the n-type material, they will be capable of crossing the barrier into the p-type material—and current will flow. This additional energy can be supplied by applying a positive voltage to the p-type material, as shown in the figure. The negatively charged electrons will then be highly attracted to the positive voltage across the junction. 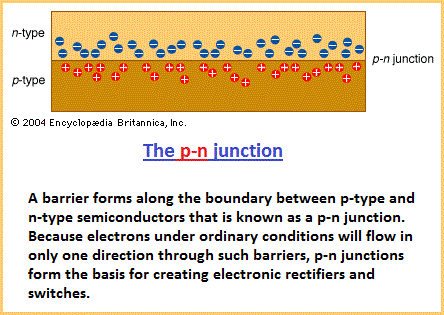 A p-n junction that conducts electricity when energy is added to the n material is called forward-biased because the electrons move forward into the holes. If voltage is applied in the opposite direction—a positive voltage connected to the n side of the junction—no current will flow. The electrons in the n material will still be attracted to the positive voltage, but the voltage will now be on the same side of the barrier as the electrons. In this state a junction is said to be reverse-biased. Since p-n junctions conduct electricity in only one direction, they are a type of diode. Diodes are essential building blocks of semiconductor switches. 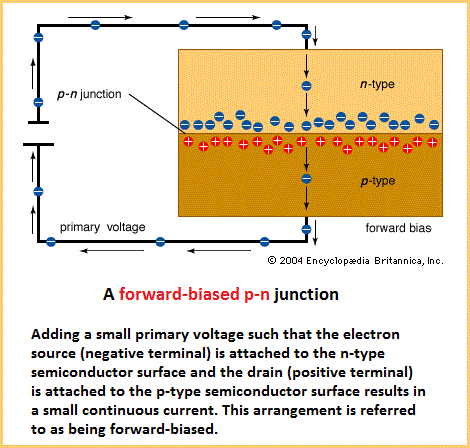 All sorts of devices, such as diodes, transistors, capacitors, and resistors, can be built with p- and n-type semiconductors. It is convenient to be able to manufacture all of these different electronic components from the same few basic manufacturing steps. ICs are made of layers, from about 0.000005 to 0.1 mm thick, that are built on the semiconductor substrate one layer at a time, with perhaps 30 or more layers in a final chip. Creating the different electrical components on a chip is a matter of outlining exactly where areas of n- and p-type are to be located on each layer. Each layer is etched, using lines and geometric shapes in the exact locations where the material is to be deposited. Different colours represent different layers. A wafer can be changed in one of three fundamental ways: by deposition (that is, adding a layer), by etching or removing a layer, or by implantation (altering a layer's composition). Source: "Integrated Circuit (IC)." Encyclopædia Britannica Ultimate Reference Suite, 2013. |
Let's provide some definitions to a few terms used in electronics:
|
Prior to the invention of the bipolar transistor in 1947, semiconductors were used only as two-terminal devices, such as rectifiers and photodiodes. During the early 1950s, germanium was the major semiconductor material. However, it proved unsuitable for many applications, because devices made of the material exhibited high leakage currents at only moderately elevated temperatures. Since the early 1960s, silicon has become a practical substitute, virtually supplanting germanium as a material for semiconductor fabrication. The main reasons for this are twofold: (1) silicon devices exhibit much lower leakage currents, and (2) high-quality silicon dioxide (SiO2), which is an insulator, is easy to produce. Silicon technology is now by far the most advanced among all semiconductor technologies, and silicon-based devices constitute more than 95 percent of all semiconductor hardware sold worldwide. Many of the compound semiconductors have electrical and optical properties that are absent in silicon. These semiconductors, especially gallium arsenide, are used mainly for high-speed and optoelectronic applications. (A Capacitor is a) device for storing electrical energy, consisting of two conductors in close proximity and insulated from each other. A simple example of such a storage device is the parallel-plate capacitor. If positive charges with total charge +Q are deposited on one of the conductors and an equal amount of negative charge -Q is deposited on the second conductor, the capacitor is said to have a charge Q. 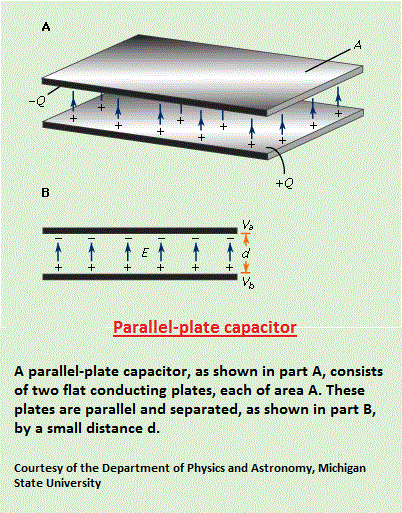 Capacitors have many important applications. They are used, for example, in digital circuits so that information stored in large computer memories is not lost during a momentary electric power failure; the electric energy stored in such capacitors maintains the information during the temporary loss of power. Capacitors play an even more important role as filters to divert spurious electric signals and thereby prevent damage to sensitive components and circuits caused by electric surges. Source: "Capacitor." Encyclopædia Britannica Ultimate Reference Suite, 2013. |
|
Two terminal (di-ode) junction devices The p-i-n diodeA p-i-n diode is a p-n junction with an impurity profile tailored so that an intrinsic layer, the “i region,” is sandwiched between a p layer and an n layer. The p-i-n diode has found wide application in microwave circuits. It can be used as a microwave switch with essentially constant depletion-layer capacitance (equal to that of a parallel-plate capacitor having a distance between the plates equal to the i-region thickness) and high power-handling capability. A p-n junction diode is a solid-state device that has two terminals. Depending on impurity distribution, device geometry, and biasing condition, a junction diode can perform various functions. There are more than 50,000 types of diodes with voltage ratings from less than 1 volt to more than 2,000 volts and current ratings from less than 1 milliampere to more than 5,000 amperes. A p-n junction also can generate and detect light and convert optical radiation into electrical energy. Rectifier This type of p-n junction diode is specifically designed to rectify an alternating current—i.e., to give a low resistance to current flow in one direction and a very high resistance in the other direction. Such diodes are generally designed for use as power-rectifying devices that operate at frequencies from 50 hertz to 50 kilohertz. The majority of rectifiers have power-dissipation capabilities from 0.1 to 10 watts and a reverse breakdown voltage from 50 to more than 5,000 volts. (A high-voltage rectifier is made from two or more p-n junctions connected in series.) Source: "Semiconductor Device." Encyclopædia Britannica Ultimate Reference Suite, 2013. |
|
(A transistor is a) semiconductor device for amplifying, controlling, and generating electrical signals. Transistors are the active components of integrated circuits, or “microchips,” which often contain millions of these minuscule devices etched into their shiny surfaces. Deeply embedded in almost everything electronic, transistors have become the nerve cells of the Information Age. The transistor was invented in 1947–48 by three American physicists, John Bardeen, Walter H. Brattain, and William B. Shockley, at the American Telephone and Telegraph Company's Bell Laboratories. The transistor proved to be a viable alternative to the electron tube and, by the late 1950s, supplanted the latter in many applications.
The transistor's small size, low heat generation, high reliability, and low power consumption made possible a breakthrough in the miniaturization of complex circuitry. During the 1960s and '70s, transistors were incorporated into integrated circuits, in which a multitude of components (e.g., diodes, resistors, and capacitors) are formed on a single “chip” of semiconductor material. There are typically three electrical leads in a transistor, called the emitter, the collector, and the base—or, in modern switching applications, the source, the drain, and the gate (see figure). An electrical signal applied to the base (or gate) influences the semiconductor material's ability to conduct electrical current, which flows between the emitter (or source) and collector (or drain) in most applications. A voltage source such as a battery drives the current, while the rate of current flow through the transistor at any given moment is governed by an input signal at the gate—much as a faucet valve is used to regulate the flow of water through a garden hose. 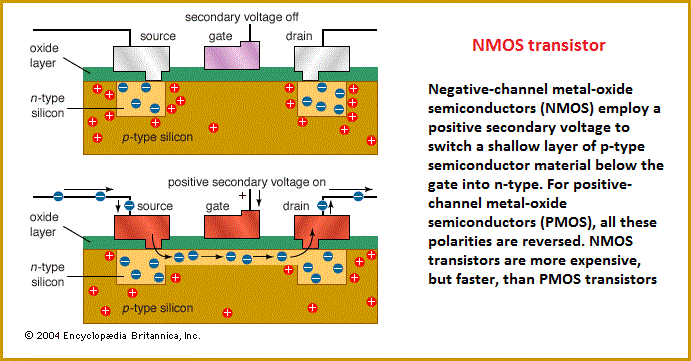 The first commercial applications for transistors were for hearing aids and “pocket” radios during the 1950s. With their small size and low power consumption, transistors were desirable substitutes for the vacuum tubes (known as “valves” in Great Britain) then used to amplify weak electrical signals and produce audible sounds. Transistors also began to replace vacuum tubes in the oscillator circuits used to generate radio signals, especially after specialized structures were developed to handle the higher frequencies and power levels involved. Low-frequency, high-power applications, such as power-supply inverters that convert alternating current (AC) into direct current (DC), have also been transistorized. Some power transistors can now handle currents of hundreds of amperes at electric potentials over a thousand volts. By far the most common application of transistors today is for computer memory chips—including solid-state multimedia storage devices for electronic games, cameras, and MP3 players—and microprocessors, where millions of components are embedded in a single integrated circuit. Here the voltage applied to the gate electrode, generally a few volts or less, determines whether current can flow from the transistor's source to its drain. In this case the transistor operates as a switch: if a current flows, the circuit involved is on, and if not, it is off. These two distinct states, the only possibilities in such a circuit, correspond respectively to the binary 1s and 0s employed in digital computers. Similar applications of transistors occur in the complex switching circuits used throughout modern telecommunications systems. The potential switching speeds of these transistors now exceed a gigahertz, or more than a billion on-and-off cycles per second. Source: "Transistor." Encyclopædia Britannica Ultimate Reference Suite, 2013. |
|
(A resistor is an) electrical component that opposes the flow of either direct or alternating current, employed to protect, operate, or control the circuit. Voltages can be divided with the use of resistors, and in combination with other components resistors can be used to make electrical waves into shapes most suited for the electrical designer's requirements. Resistors can have a fixed value of resistance, or they can be made variable or adjustable within a certain range, in which case they may be called rheostats, or potentiometers. Source: "Resistor." Encyclopædia Britannica Ultimate Reference Suite, 2013. |
|
(A Rheostat is an) adjustable resistor used in applications that require the adjustment of current or the varying of resistance in an electric circuit. The rheostat can adjust generator characteristics, dim lights, and start or control the speed of motors. Its resistance element can be a metal wire or ribbon, carbon, or a conducting liquid, depending on the application. For average currents, the metallic type is most common; for very small currents, the carbon type is used; and for large currents the electrolytic type, in which electrodes are placed in a conducting fluid, is most suitable. A special type of rheostat is the potentiometer, an instrument that measures an unknown voltage or potential difference by balancing it, wholly or in part, by a known potential difference. A more common potentiometer is simply a resistor with two fixed terminals and a third terminal connected to a variable contact arm; it is used for such purposes as a volume control in audio equipment. Source: "Rheostat." Encyclopædia Britannica Ultimate Reference Suite, 2013. |
|
Superconductivity is the total disappearance of electrical resistance below a definite temperature called the transition temperature. Because niobium has the highest transition temperature (9.3 K [-264° C, or -443° F]), among metals, niobium alloys are the most practical choice for superconducting applications. Niobium-titanium, niobium-zirconium, and niobium-tin alloys are indispensable for the application of superconductivity in machinery, magnetically levitated trains, high-energy physics, and magnetic resonance imaging. Source: "Niobium processing." Encyclopædia Britannica Ultimate Reference Suite, 2013. |
|
Discovery and composition of high-temperature superconductors Ever since Kamerlingh Onnes discovered that mercury becomes superconducting at temperatures less than 4 K, scientists have been searching for superconducting materials with higher transition temperatures. Until 1986 a compound of niobium and germanium (Nb3Ge) had the highest known transition temperature, 23 K, less than a 20-degree increase in 75 years. Most researchers expected that the next increase in transition temperature would be found in a similar metallic alloy and that the rise would be only one or two degrees. In 1986, however, the Swiss physicist Karl Alex Müller and his West German associate, Johannes Georg Bednorz, discovered, after a three-year search among metal oxides, a material that had an unprecedentedly high transition temperature of about 30 K. They were awarded the Nobel Prize for Physics in 1987, and their discovery immediately stimulated groups of investigators in China, Japan, and the United States to produce superconducting oxides with even higher transition temperatures. These high-temperature superconductors are ceramics. They contain lanthanum, yttrium, or another of the rare-earth elements or bismuth or thallium; usually barium or strontium (both alkaline-earth elements); copper; and oxygen. Other atomic species can sometimes be introduced by chemical substitution while retaining the high-Tc properties. The Table lists the member of each major family of high-Tc materials with the highest observed superconducting transition temperature. The value 134 K is the highest known Tc value. Within each family of high-Tc materials, only the subscripts (i.e., stoichiometry) vary from one compound to another. Samples in the families containing bismuth or thallium always exhibit a great deal of atomic disorder, with atoms in the “wrong” crystallographic sites and with impurity phases. It is possible that such disorder is required to make these compounds thermodynamically stable. Structures and properties The compounds have crystal structures containing planes of Cu (copper) and O (oxygen) atoms, and some also have chains of Cu and O atoms. The roles played by these planes and chains have come under intense investigation. Varying the oxygen content or the heat treatment of the materials dramatically changes their transition temperatures, critical magnetic fields, and other properties. Single crystals of the high-temperature superconductors are very anisotropic—i.e., their properties associated with a direction, such as the critical fields or the critical current density, are highly dependent on the angle between that direction and the rows of atoms in the crystal. If the number of superconducting electrons per unit volume is locally disturbed by an applied force (typically electric or magnetic), this disturbance propagates for a certain distance in the material; the distance is called the superconducting coherence length (or Ginzburg-Landau coherence length), ξ. If a material has a superconducting region and a normal region, many of the superconducting properties disappear gradually—over a distance ξ—upon traveling from the former to the latter region. In the pure (i.e., undoped) classic superconductors ξ is on the order of a few thousand angstroms, but in the high-Tc superconductors it is on the order of 1 to 10 angstroms. The small size of ξ affects the thermodynamic and electromagnetic properties of the high-Tc superconductors. For example, it is responsible for the cusp shape of the specific heat curve near Tc that was mentioned above. It is also responsible for the ability of the high-Tc superconductors to remain superconducting in extraordinarily large fields—on the order of 1,000,000 gauss (100 teslas)—at low temperatures. The high-Tc superconductors are type II superconductors. They exhibit zero resistance, strong diamagnetism, the Meissner effect, magnetic flux quantization, the Josephson effects, an electromagnetic penetration depth, an energy gap for the superconducting electrons, and the characteristic temperature dependencies of the specific heat and the thermal conductivity that are described above. Therefore, it is clear that the conduction electrons in these materials form the Cooper pairs used to explain superconductivity in the BCS theory. Thus, the central conclusions of the BCS theory are demonstrated. Indeed, that theory guided Bednorz and Müller in their search for high-temperature superconductors. It is not known, however, why the transition temperatures of these oxides are so high. It was generally believed that the members of a Cooper pair are bound together because of interactions between the electrons and the lattice vibrations (phonons), but it is unlikely that these interactions are strong enough to explain transition temperatures as high as 90 K. Most experts believe that interactions among the electrons generate high-temperature superconductivity. The details of this interaction are difficult to treat theoretically because the motions of the electrons are strongly correlated with each other and because magnetic phenomena play an important part in determining the microscopic properties of these materials. These strong correlations and magnetic properties may be responsible for unusual temperature dependencies of the electric resistivity ρ and Hall coefficient RH in the normal state (i.e., above Tc). It is observed that at temperatures above Tc the electric resistivity, although higher for superconductors than for typical metals in the normal state, is roughly proportional to the temperature T, an unusually weak temperature dependence. Measurements of RH show it to be significantly temperature-dependent in the normal state (sometimes proportional to 1/T) rather than being roughly independent of T, which is the case for ordinary materials. 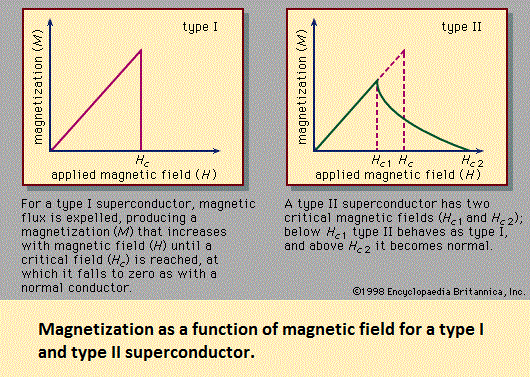 Source: "Superconductivity." Encyclopædia Britannica Ultimate Reference Suite, 2013. |
Subject page first Originated (saved into a folder): Thursday, November 13, 2014... 5:50 AM
Page re-Originated: Sunday, 24-Jan-2016... 08:51 AM
Initial Posting: Saturday, 13-Feb-2016... 10:59 AM
Updated Posting: Monday, 24-June-2019... 10:55 AM
Herb O. Buckland
herbobuckland@hotmail.com